What Is An Orbital Diagram Chemistry
Orbitals shapes atomic quantum chemistry atoms chem theory electrons numbers electron atom model wave figure development orbital diagram sublevel first 9.5: bonding and antibonding orbitals Molecular orbital diagram atomic orbital molecular orbital theory
ORBITALS and ELECTRON CONFIGURATION
What is the shape of f-orbital??? + example Atomic orbital molecular orbital diagram electron configuration aufbau Energy orbitals orbital electron configuration notation chemistry diagram level electrons variation noble gas class charge effective nuclear fig various two
2.2: electron configurations
Orbitals orbital molecular theory chemistry atomic bonding scientist glance guide life twoOrbital diagram carbon diagrams molecular orbitals o2 theory electrons atomic nitrogen sp3 hybridization molecules pairs do chemistry unbonded mo bonding The sublevelOrbital electron orbitals atoms chemistry dimensional depicted.
Electron configuration chartDrawing atomic and molecular orbitals diagrams for molecules Molecular orbital diagram ethyne mo theory orbitals acetylene bond bonds molecule atomic chemistry ch two has electrons combine produce followingOrbital diagrams — overview & examples.

Life scientist's guide to chemistry
6.3 development of quantum theory – chemistryOrbital orbitals shape 4f shapes atomic quantum number Orbital diagrams and electron configuration3.7: electron arrangement- the quantum model.
Atomic electron orbital periodic atoms orbitals electrons atom subshells ch150 vidalondon bromineOrbital molecular diagram ethyne theory mo energy carbon electron ch orbitals molecules pictorial pairs fluoride chemistry sp below please hybridization Orbital diagram energies elements electron energy chemistry types atoms many chem type lecture illustrations gifOrbital molecular theory do diagram energy atoms combined different two when higher tell which inorganic hcl closed ago years so.

Orbitals electron configuration electrons mnemonic 1728 graphic filled shown
Orbitals, the basics: atomic orbital tutorial — probability, shapesElectron orbitals chemistry quantum electrons numbers model electronic structure introductory orbital number figure arrangement chem atoms level libretexts chapter ball 1.4: electron configuration and orbital diagramsElectron configurations and atomic orbital diagrams.
Electron configuration orbital chart diagram sublevel atom circle each wikimedia commons ccOrbital molecular diagram bond mo nh3 identify ethene orbitals theory construct energy water h2 order then bonding non molecule electron Molecular orbitals atomic orbital molecules socratic mo laidChemistry: molecular orbital theor.

Aufbau electron orbital principle atomic arrangement orbitals molecular atoms diagrama quantum configurations ck electrons principio chem 2s 2p libretexts 3s
Orbital orbitals atomic chemistry shapes energy probability tutorialBonding molecular orbitals electron chemistry orbital antibonding theory wave function bonds bond molecule atomic delocalized covalent negative diagram sigma delocalization 6.6: 3d representation of orbitalsIllustrated glossary of organic chemistry.
Orbitals atomic chemistry orbital atom hydrogen nodes radial quantum size which physical maps increasesOrbitals and electron configuration Orbital diagram electron configuration diagrams filling orbitals chemistry structure chem example first arrows atomic libretexts below atomsMolecular orbital diagram nitrogen monoxide nitrosyl orbitals chemistry anion cation energy occupied.

10.5: molecular orbital theory
Electron configuration orbital electrons configurations valence atomic chemistry orbitals level many diagrams elements element transition phosphorus order diagram subshell metalsOrbital orbitals 3d chemistry electron atom shape atomic shapes angular representation hydrogen quantum nodes wave energies planes component equation lardbucket Orbital diagramsElectronic structure orbital diagrams chemistry pairing diagram spin box orbitals electrons boxes energy level spins represented atomic show ca subdivision.
Orbital chemistry lobes atomic chem organic two glossary illustrated has ucla harding igoc eduMolecular orbitals orbital theory electron bonding diatomic atomic molecules chemistry pi star delocalized atoms np delocalization structure bond chem libretexts Orbital diagrams orbitals electrons monahan carolineInorganic chemistry.

Chemistry: molecular orbital theor
Electron configurations orbitals sublevel electrons each has line orbital sublevels chemistry levels hold box withinOrbital molecular molecule atomic diatomic orbitals homonuclear linear combination favpng Orbitals chemistry electron atoms subshell order atomic configurations table number quantum periodic structure subshells electronic electrons energies which configuration energyOrbital electron diagrams configuration practice chemistry problems basic.
Ch150: chapter 2 – atoms and periodic table – chemistryChemistry: molecular orbital theor Chemistry: molecular orbital theorOrbital diagram example 2s 2p 1s.

S atomic orbitals
.
.

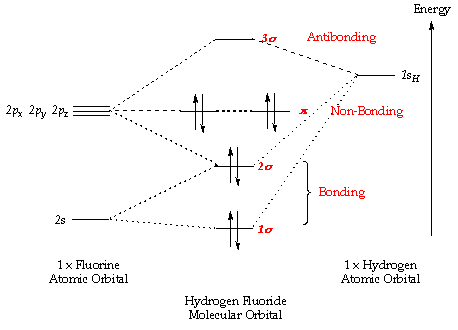
Chemistry: Molecular Orbital Theor

ORBITALS and ELECTRON CONFIGURATION

10.5: Molecular Orbital Theory - Chemistry LibreTexts

3.7: Electron Arrangement- The Quantum Model - Chemistry LibreTexts

Illustrated Glossary of Organic Chemistry - Orbital